Abstract
Review Article
New insights from cardiac muscle applied to skeletal muscle
Gerry A Smith*
Published: 15 January, 2021 | Volume 6 - Issue 1 | Pages: 007-013
I have recently described the origin of the second Ca2+ binding in the triggering of contractile activity in cardiac myofibrils that is the origin of the Ca2+ Hill coefficient of 2 for the ATPase. This site is not a simple protein binding site and cannot be measured by 45Ca2+ binding. The myofibril protein unit requirements are described by me and so are the consequences of disruption of the function of these units and the related medical outcomes. The purpose of this paper is to review the topic and extend the reasoning to the function of skeletal muscle and cite the literature that supports this.
Read Full Article HTML DOI: 10.29328/journal.jccm.1001109 Cite this Article Read Full Article PDF
References
- Smith GA, Vandenberg JI, Freestone NS, Dixon HBF. The effect of Mg2+ on cardiac muscle function: Is CaATP the substrate for priming myofibril cross-bridge formation and Ca2+ reuptake by the sarcoplasmic reticulum? Biochem J. 2001; 354: 539-551. PubMed: https://pubmed.ncbi.nlm.nih.gov/11237858/
- Smith GA. Calcium, actomyosin kinetics, myosin binding protein-c and hypertrophic cardiomyopathy. J Integr Cardiol. 2019; 5: 1-2.
- Smith GA. The Mechanisms of the Frank-Starling Law and Familial Cardiomyopathy are Different. The Function of Myosin Binding Protein-C is Retained on Myocyte Length Increase and Force Generated is Kinase controlled. J Integr Cardiol. 2019; 5: 1-3.
- Smith GA. Angiotensin II type 1 receptor and the activation of Myosin Light-Chain Kinase and Protein Kinase C-βII: Mini Review. J Cardiol Cardiovasc Med. 2020; 5: 024-028. https://www.heighpubs.org/jccm/jccm-aid1081.php
- Smith GA. The Mechanisms of Cardiac Myopathies, a kinetics approach. Leading review. J Cardiol Cardiovasc Med. 2020; 5: 141-152. https://www.heighpubs.org/jccm/jccm-aid1101.php
- Brandt PW, Diamond MS, Gluck B, Kawai M, Schachat F. Molecular Basis of Cooperativity in Vertebrate Muscle Thin Filaments Carlsberg Res. Commun. 1984; 49: 155-167.
- Hofmann PA, Hartzell HC, Moss RL. Alterations in Ca2+ sensitive tension due to partial extraction of C-protein from rat skinned cardiac myocytes and rabbit skeletal muscle fibers. J Gen Physiol. 1991; 97: 1141-1163. PubMed: https://pubmed.ncbi.nlm.nih.gov/1678777/
- Katrukha IA. Human Cardiac Troponin Complex. Structure and Functions. Biochemistry (Moscow). 2013; 78: 1447-14650. PubMed: https://pubmed.ncbi.nlm.nih.gov/24490734/
- Layland J, Solaro RJ, Shah AM. Regulation of cardiac contractile function by troponin I phosphorylation. Cardiovascular Res. 2005; 66: 12–21. PubMed: https://pubmed.ncbi.nlm.nih.gov/15769444/
- Westfall MV, Lee AM, Robinson DA. Differential Contribution of Troponin I Phosphorylation Sites to the Endothelin-modulated Contractile Response. J Biol Chem. 2005; 280: 41324–41331. PubMed: https://pubmed.ncbi.nlm.nih.gov/16236710/
- Sheng JJ, Jin JP. TNNI1, TNNI2 and TNNI3: Evolution, regulation, and protein structure–function relationships. Gene. 2016; 576: 385-394. PubMed: https://pubmed.ncbi.nlm.nih.gov/26526134/
- Robinson P, Lipscomb S, Preston LC, Altin E, Watkins H, et al. Mutations in fast skeletal troponin I, troponin T, and beta-tropomyosin that cause distal arthrogryposis all increase contractile function. FASEB J. 2007; 21: 896–905. PubMed: https://pubmed.ncbi.nlm.nih.gov/17194691/
- Seidman JG, Seidman C. The genetic basis for cardiomyopathy: from mutation identification to mechanistic paradigms. Cell. 2001; 104: 557–567. PubMed: https://pubmed.ncbi.nlm.nih.gov/11239412/
- Curila K, Benesova L, Penicka M, Minarik M, Zemanek D, et al. Spectrum and clinical manifestations of mutations in genes responsible for hypertrophic cardiomyopathy. Acta Cardiol. 2012; 67: 23–29. PubMed: https://pubmed.ncbi.nlm.nih.gov/22455086/
- Heling LWHJ, Geeves MA, Kad NM. MyBP-C: one protein to govern them all. J. Muscle Res. and Cell Motility. 2020; 41: 91–101.
- Lin B, Govindan S, Lee K, Zhao P, Renzhi Han R, et al. Cardiac Myosin Binding Protein-C Plays No Regulatory Role in Skeletal Muscle Structure and Function. PLoS One. 2013; 8: e69671. PubMed: https://pubmed.ncbi.nlm.nih.gov/23936073/
- Lee RS. Effect of the Ca2+ Binding Properties of Troponin C On Skeletal and Cardiac Muscle Force Development. Thesis the Ohio State University. 2010.
- Potter JD, Gergely J. J BiolChem. 1975; 250: 4628-4633.
- Kretsinger RH. CRC Crit Rev Biochem. 1980; 8: 119-174.
- Sia SK, Li MX, Spyracopoulos L, Gagne SM, Liu W, et al. J Biol Chem. 1997; 272: 18216-18221.
- Brandt PW, Cox RN, Kawai M. Can the binding of Ca2+ to two regulatory sites on troponin-C determine the steep pCa/tension relationship of skeletal muscle? Proc Natl Acad Sci. 1980; 77: 4717-4720. PubMed: https://pubmed.ncbi.nlm.nih.gov/6933518/
- Kampourakis T, Yan Z, Gautel M, Sun YB, Irving M. Myosin binding protein-C activates thin filaments and inhibits thick filaments in heart muscle cells PNAS. 2014; 111: 18763–18768. PubMed: https://pubmed.ncbi.nlm.nih.gov/25512492/
- Flashman E, Watkins H, Redwood C. Localization of the binding site of the C-terminal domain of cardiac myosin-binding protein-C on the myosin rod. Biochem J. 2007; 401: 97–102. PubMed: https://pubmed.ncbi.nlm.nih.gov/16918501/
- van Dijk SJ. Bezold KL, Harris SP. Earning Stripes: Myosin Binding Protein-C Interactions with Actin. Pflugers Arch. 2014; 466: 445–450. PubMed: https://pubmed.ncbi.nlm.nih.gov/24442149/
- Lu Y, Kwan AH, Trewhella J, Jeffries CM. Communication: The C0C1 Fragment of Human Cardiac Myosin Binding Protein C Has Common Binding Determinants for Both Actin and Myosin. J Mol Biol. 2011; 413: 908–913. PubMed: https://pubmed.ncbi.nlm.nih.gov/21978665/
- Moss RL, Giulian GG, Greaser ML. The effects of partial extraction of TnC upon the tension-pCa relationship in rabbit skinned skeletal muscle fibers. J Gen Physiol. 1985; 86: 585–600. PubMed: https://pubmed.ncbi.nlm.nih.gov/4056736/
- Morimoto S, Ohtsuki I. Role of Troponin C in Determining the Ca2+ -Sensitivity and Cooperativity of the Tension Development in Rabbit Skeletal and Cardiac Muscles. J Biochem. 1994; 115: 144-146.
- Zot HG, Potter JD. A structural role for the Ca2+-Mg2+ sites on troponin C in the regulation of muscle contraction. Preparation and properties of troponin C depleted myofibrils. J Biol Chem. 1982; 257: 7678-7683. PubMed: https://pubmed.ncbi.nlm.nih.gov/6211445/
- Pohlmann L, Kröger I, Vignier N, Schlossarek S, Krämer E, et al. Cardiac Myosin-Binding Protein C Is Required for Complete Relaxation in Intact Myocytes. Circ Res. 2007; 101: 928-938. PubMed: https://pubmed.ncbi.nlm.nih.gov/17823372/
- Lecarpentier Y, Vignier N, Oliviero P, Guellich A, Carrier L, et al. Cardiac Myosin-Binding Protein C Modulates the Tuning of the Molecular Motor in the Heart. Biophysical J. 2008; 95: 720–728. PubMed: https://pubmed.ncbi.nlm.nih.gov/18375505/
- Razumova MV, Bezold KL, Tu AY, Regnier M, Harris SP. Contribution of the Myosin Binding Protein C Motif to Functional Effects in Permeabilized Rat Trabeculae. J Gen Physiol. 2008; 132: 575–585. PubMed: https://pubmed.ncbi.nlm.nih.gov/18955596/
- Lin BL, Li A, Mun JY, Previs MJ, Previs SB, et al. Skeletal myosin binding protein-C isoforms regulate thin filament activity in a Ca2+-dependent manner. Nature, Scientific Reports. 2018; 8: 2604. PubMed: https://pubmed.ncbi.nlm.nih.gov/29422607/
- Oakley CE, Chamoun J, Brown LJ, Hamblya BD. Binding protein-C: Enigmatic regulator of cardiac contraction. Int J Biochem Cell Biol. 2007; 39: 2161–2166. PubMed: https://pubmed.ncbi.nlm.nih.gov/17320463/
- Orlova A, Galkin VE, Cy MJ, Jeffries Cy MJ, Egelman EH, et al. The N-terminal Domains of Myosin Binding Protein C Can Bind Polymorphically to F-Actin. J Mol Biol. 2011; 412: 379–386. PubMed: https://pubmed.ncbi.nlm.nih.gov/21821050/
- Li A. Nelson SR, Rahmanseresht S, Braet F, Cornachione AS, et al. Skeletal MyBP-C isoforms tune the molecular contractility of divergent skeletal muscle systems. PNAS. 2019; 116: 21882–21892. PubMed: https://pubmed.ncbi.nlm.nih.gov/31591218/
- Chen Z, Zhao TJ, Li J, Gao YS, Meng FG, et al. Slow skeletal muscle myosin-binding protein-C (MyBPC1) mediates recruitment of muscle-type creatine kinase (CK) to myosin. Biochem J. 2011; 436: 437-445. PubMed: https://pubmed.ncbi.nlm.nih.gov/21426302/
Figures:
Figure 1

Figure 2
Figure 3
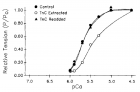
Figure 4

Figure 5

Figure 6
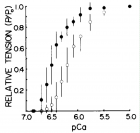
Figure 7
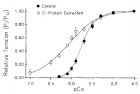
Figure 8
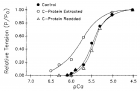
Figure 9
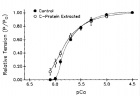
Figure 10
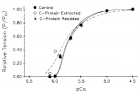
Figure 11
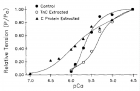
Figure 12
Figure 13
Similar Articles
-
Left Atrial Remodeling is Associated with Left Ventricular Remodeling in Patients with Reperfused Acute Myocardial InfarctionChristodoulos E. Papadopoulos*,Dimitrios G. Zioutas,Panagiotis Charalambidis,Aristi Boulbou,Konstantinos Triantafyllou,Konstantinos Baltoumas,Haralambos I. Karvounis,Vassilios Vassilikos. Left Atrial Remodeling is Associated with Left Ventricular Remodeling in Patients with Reperfused Acute Myocardial Infarction. . 2016 doi: 10.29328/journal.jccm.1001001; 1: 001-008
-
Mid-Ventricular Ballooning in Atherosclerotic and Non-Atherosclerotic Abnormalities of the Left Anterior Descending Coronary ArteryStefan Peters*. Mid-Ventricular Ballooning in Atherosclerotic and Non-Atherosclerotic Abnormalities of the Left Anterior Descending Coronary Artery. . 2016 doi: 10.29328/journal.jccm.1001002; 1:
-
Concentration Polarization of Ox-LDL and Its Effect on Cell Proliferation and Apoptosis in Human Endothelial CellsShijie Liu*,Jawahar L Mehta,Yubo Fan,Xiaoyan Deng,Zufeng Ding*. Concentration Polarization of Ox-LDL and Its Effect on Cell Proliferation and Apoptosis in Human Endothelial Cells. . 2016 doi: 10.29328/journal.jccm.1001003; 1:
-
Intermittent Left Bundle Branch Block: What is the Mechanism?Hussam Ali*,Riccardo Cappato. Intermittent Left Bundle Branch Block: What is the Mechanism?. . 2017 doi: 10.29328/journal.jccm.1001004; 2:
-
Congenital Quadricuspid Aortic Valve, a Rare Cause of Aortic Insufficiency in Adults: Case ReportCyrus Kocherla*,Kalgi Modi. Congenital Quadricuspid Aortic Valve, a Rare Cause of Aortic Insufficiency in Adults: Case Report. . 2017 doi: 10.29328/journal.jccm.1001005; 2: 003-007
-
Short and Medium-Term Evaluation of Patients in Coronary Post-Angioplasty: Préliminary results at the Cardiology Department of the Hospital University Aristide Le Dantec of Dakar (Senegal): Study on 38 CasesDioum M*,Aw F,Masmoudi K,Gaye ND,Sarr SA,Ndao SCT, Mingou J,Ngaidé AA,Diack B,Bodian M,Ndiaye MB,Diao M,Ba SA. Short and Medium-Term Evaluation of Patients in Coronary Post-Angioplasty: Préliminary results at the Cardiology Department of the Hospital University Aristide Le Dantec of Dakar (Senegal): Study on 38 Cases. . 2017 doi: 10.29328/journal.jccm.1001006; 2: 008-012
-
Indications and Results of Coronarography in Senegalese Diabetic Patients: About 45 CasesNdao SCT*,Gaye ND,Dioum M,Ngaide AA,Mingou JS,Ndiaye MB, Diao M,Ba SA. Indications and Results of Coronarography in Senegalese Diabetic Patients: About 45 Cases. . 2017 doi: 10.29328/journal.jccm.1001007; 2: 013-019
-
Procedure utilization, latency and mortality: Weekend versus Weekday admission for Myocardial InfarctionNader Makki,David M Kline,Arun Kanmanthareddy,Hansie Mathelier,Satya Shreenivas,Scott M Lilly*. Procedure utilization, latency and mortality: Weekend versus Weekday admission for Myocardial Infarction. . 2017 doi: 10.29328/journal.jccm.1001008; 2: 020-025
-
Spontaneous rupture of a giant Coronary Artery Aneurysm after acute Myocardial InfarctionOğuzhan Çelik,Mucahit Yetim,Tolga Doğan,Lütfü Bekar,Macit Kalçık*,Yusuf Karavelioğlu. Spontaneous rupture of a giant Coronary Artery Aneurysm after acute Myocardial Infarction. . 2017 doi: 10.29328/journal.jccm.1001009; 2: 026-028
-
Thrombolysis, the only Optimally Rapid Reperfusion TreatmentVictor Gurewich*. Thrombolysis, the only Optimally Rapid Reperfusion Treatment. . 2017 doi: 10.29328/journal.jccm.1001010; 2: 029-034
Recently Viewed
-
Exophthalmos Revealing a Spheno Temporo Orbital MeningiomaHassina S*, Krichene MA, Hazil Z, Bekkar B, Hasnaoui I, Robbana L, Bardi S, Akkanour Y, Serghini L, Abdallah EL. Exophthalmos Revealing a Spheno Temporo Orbital Meningioma. Int J Clin Exp Ophthalmol. 2024: doi: 10.29328/journal.ijceo.1001055; 8: 001-003
-
Value of electrocardiographic T wave inversion in lead aVL in prediction of Mid Left Anterior Descending Stenosis in patients with stable Coronary Artery diseaseGehan Magdy*,Awad Yousef. Value of electrocardiographic T wave inversion in lead aVL in prediction of Mid Left Anterior Descending Stenosis in patients with stable Coronary Artery disease. J Cardiol Cardiovasc Med. 2017: doi: 10.29328/journal.jccm.1001015; 2: 056-062
-
Calciphylaxis in HemodialysisLuca Sgarabotto*, Paola Baldini Anastasio, Nicola Marchionna, Monica Zanella. Calciphylaxis in Hemodialysis. J Clini Nephrol. 2023: doi: 10.29328/journal.jcn.1001107; 7: 044-046
-
Metabolic syndrome: A case reportDragan Klaric,Marta Martinis*,Marta Klaric. Metabolic syndrome: A case report. Ann Clin Endocrinol Metabol. 2021: doi: 10.29328/journal.acem.1001022; 5: 031-035
-
Utilization of Molecular Simulation Applications for Stability Testing of Anthocyanin Compounds in Black ElderberryRichard Benedict*. Utilization of Molecular Simulation Applications for Stability Testing of Anthocyanin Compounds in Black Elderberry. Arch Biotechnol Biomed. 2023: doi: 10.29328/journal.abb.1001035; 7: 012-017
Most Viewed
-
Evaluation of Biostimulants Based on Recovered Protein Hydrolysates from Animal By-products as Plant Growth EnhancersH Pérez-Aguilar*, M Lacruz-Asaro, F Arán-Ais. Evaluation of Biostimulants Based on Recovered Protein Hydrolysates from Animal By-products as Plant Growth Enhancers. J Plant Sci Phytopathol. 2023 doi: 10.29328/journal.jpsp.1001104; 7: 042-047
-
Sinonasal Myxoma Extending into the Orbit in a 4-Year Old: A Case PresentationJulian A Purrinos*, Ramzi Younis. Sinonasal Myxoma Extending into the Orbit in a 4-Year Old: A Case Presentation. Arch Case Rep. 2024 doi: 10.29328/journal.acr.1001099; 8: 075-077
-
Feasibility study of magnetic sensing for detecting single-neuron action potentialsDenis Tonini,Kai Wu,Renata Saha,Jian-Ping Wang*. Feasibility study of magnetic sensing for detecting single-neuron action potentials. Ann Biomed Sci Eng. 2022 doi: 10.29328/journal.abse.1001018; 6: 019-029
-
Pediatric Dysgerminoma: Unveiling a Rare Ovarian TumorFaten Limaiem*, Khalil Saffar, Ahmed Halouani. Pediatric Dysgerminoma: Unveiling a Rare Ovarian Tumor. Arch Case Rep. 2024 doi: 10.29328/journal.acr.1001087; 8: 010-013
-
Physical activity can change the physiological and psychological circumstances during COVID-19 pandemic: A narrative reviewKhashayar Maroufi*. Physical activity can change the physiological and psychological circumstances during COVID-19 pandemic: A narrative review. J Sports Med Ther. 2021 doi: 10.29328/journal.jsmt.1001051; 6: 001-007

HSPI: We're glad you're here. Please click "create a new Query" if you are a new visitor to our website and need further information from us.
If you are already a member of our network and need to keep track of any developments regarding a question you have already submitted, click "take me to my Query."